By studying chemical bonding, scientists can gain insights into the structure and reactivity of different substances. The type of bond formed between atoms influences various factors such as molecular geometry, bond length, and bond energy. Understanding chemical bonds is crucial for understanding the properties and behavior of molecules and compounds. Ionic compounds often have high melting and boiling points due to the strong electrostatic attractions between ions.The transfer of electrons results in the formation of cations ( positively charged ions) and anions ( negatively charged ions).Ionic compounds exist as a lattice structure, where positive and negative ions are held together by electrostatic forces.They are typically formed between a metal and a nonmetal atom.Ionic bonds involve the transfer of electrons from one atom to another.In non polar covalent bonds, the electron density is evenly distributed, and there is no significant charge separation. In polar covalent bonds, there is an uneven distribution of electron density, resulting in a partial positive and partial negative charge on the atoms.Covalent bonds can be further classified as polar or nonpolar depending on the electronegativity difference between the atoms involved.They are typically formed between nonmetal atoms.Covalent bonds involve the sharing of electrons between atoms.Types of Chemical BondsĬhemical bonds can be classified into two main types: covalent bonds and ionic bonds. The attraction between these oppositely charged ions forms the ionic bond. As a result, one atom becomes positively charged (cation) and the other becomes negatively charged (anion). Ionic bonds form between atoms with significantly different electronegativity values, causing one atom to attract electrons more strongly than the other. This type of bond is commonly found in ionic compounds. Ionic bonds occur when there is a transfer of electrons between atoms, resulting in the formation of ions. The carbon atom shares two electrons with each oxygen atom, resulting in a stable structure. For example, in the CO2 molecule (carbon dioxide), one carbon atom forms a covalent bond with two oxygen atoms. This type of bond is commonly found in molecular compounds. 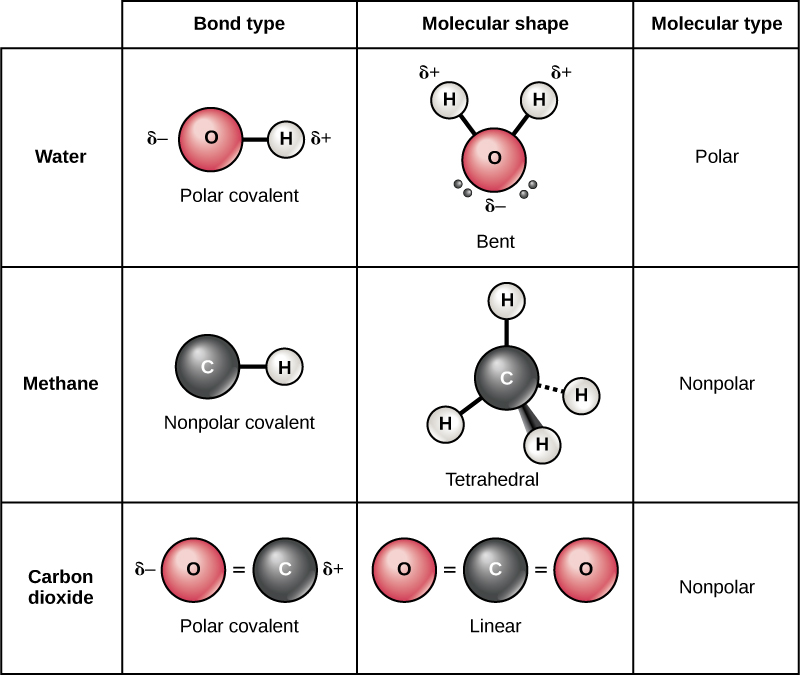
Covalent BondsĬovalent bonds occur when two or more atoms share electrons to achieve a stable electron configuration. The outermost shell of an atom, known as the valence shell, determines its chemical behavior. Atoms consist of a nucleus, which contains protons and neutrons, and electrons that orbit around the nucleus. Definition of Chemical BondsĬhemical bonds occur when atoms interact with each other by sharing or transferring electrons. In this section, we will explore the definition of chemical bonds and the different types of chemical bonds that exist. 
They are essential for the formation of all matter around us. Here is a concise table summarizing the key information about the nature of the CO2 bond: Bond Type Compound Covalent CO2 Understanding Chemical BondsĬhemical bonds are the forces that hold atoms together in a molecule or compound. Covalent bonds typically occur between nonmetal atoms, and since carbon and oxygen are both nonmetals, they form a covalent bond in CO2. This means that the atoms share electrons to form the bond, rather than transferring or gaining electrons as in an ionic bond. In the case of CO2, the carbon-oxygen bonds are covalent. When it comes to determining whether CO2 is ionic or covalent, we need to consider the nature of the bond between the atoms. Carbon dioxide (CO2) is a chemical compound that consists of one carbon atom bonded to two oxygen atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
